Acmeware Achieves 100% Submission Success
Featured article
Acmeware completes 100% successful submissions for eCQM, PQRS, Hospital IQR, and Joint Commission ORYX using OneView for acute and ambulatory settings.
The FY 2023 IPPS Proposed Rule includes major updates to the Hospital IQR Program and Medicare Promoting Interoperability Program for eligible hospitals and CAHs.
Article content
CMS recently published the FY 2023 Inpatient Prospective Payment System (IPPS) Proposed Rule which includes several proposed changes to the Hospital Inpatient Quality Reporting (IQR) Program and Medicare Promoting Interoperability Program for eligible hospitals and critical access hospitals (CAHs). For the full text of the rule, see the Federal Register.
CMS has proposed numerous changes related to the Hospital Inpatient Quality Reporting (IQR) Program including the adoption of ten new measures and the refinement of two existing measures.
In this IPPS proposed rule, CMS has proposed ten new measures with voluntary and mandatory reporting periods beginning with the CY 2023 reporting year.
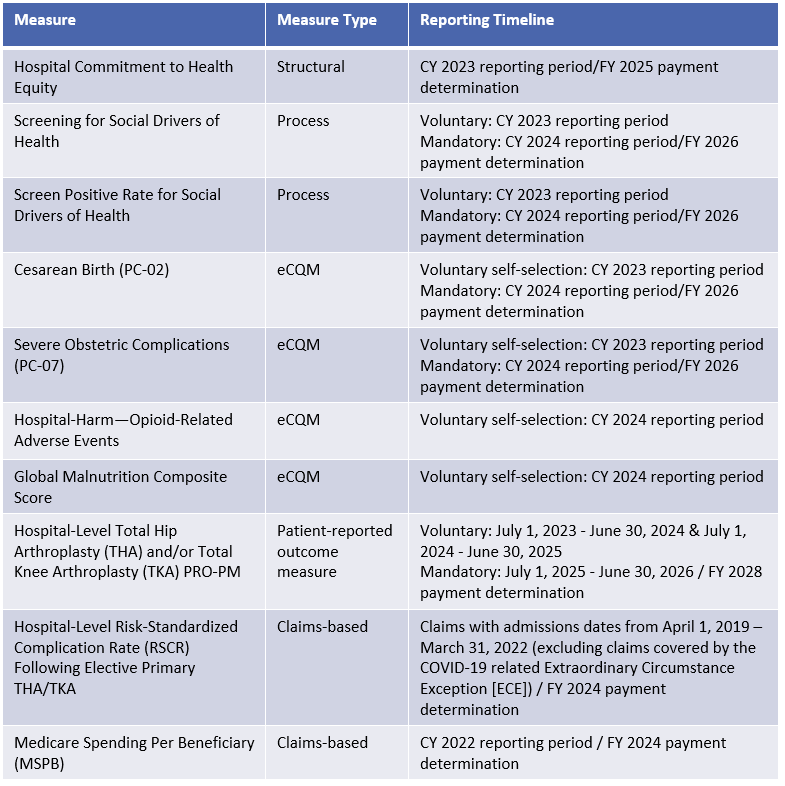
CMS is proposing refinements to two measures beginning with the FY 2024 payment determination:
CMS proposed to establish a hospital quality designation for maternity care that would be publicly reported beginning fall of 2023.
Beginning with the CY 2024 reporting period, CMS proposed to increase eCQM reporting requirements from four eCQMs to six eCQMs (three mandatory and three self-selected).
CMS proposed to modify the eCQM validation policy by increasing the submission requirement from 75% to 100% of requested medical records.
CMS proposed several changes to the PI Program beginning with the CY 2023 EHR Reporting Period:
Scoring changes include increasing Public Health and Clinical Data Exchange from 10 to 25 points, Electronic Prescribing from 10 to 20 points, reducing Health Information Exchange from 40 to 30 points, and Provide Patients Electronic Access from 40 to 25 points.
If you have questions about your hospital reporting, please contact us.